Hundreds of high-cost, low-value drugs come onto the market every year1. Some of these new high-cost drugs are just combinations of older, existing drugs. In many cases, the clinical effects of these new combinations can be easily duplicated by using their original components — at far lower cost.
Pharmacy plan sponsors can avoid unnecessary costs by spending pharmacy dollars on only the most clinically appropriate, cost-efficient medications. In this article we’ll look at muscle relaxants; a widely used, often overpriced drug category.
Skeletal muscle relaxants are widely used for acute pain and spasms associated with musculoskeletal disorders. One study estimates that up to 68 per 1,000 persons between age 18-64 in the U.S. are taking prescription skeletal muscle relaxants2. That equates to over 13.7 million people. 3
There are two distinct problems associated with the long-term use of skeletal muscle relaxants; one is clinical, the other is financial.
Clinical issues
From the clinical perspective, recommendations generally limit skeletal muscle relaxants to short-term use. Treatments may extend to a maximum of three weeks, since they have not been shown to work for muscle spasms beyond that time.4 Regardless of length of use, skeletal muscle relaxants can cause serious side effects such as falls, fracture, vehicle crashes, abuse, dependence and overdose.
However, long-term use of skeletal muscle relaxants has increased significantly. A recently published study found a 192% growth in the number of office visits resulting in continued skeletal muscle relaxant therapy in the period between 2005–2016 — nearly 25 million such visits per year. 5
This increase occurred despite research that indicates muscle relaxants may not be any more effective in managing pain than over-the-counter medications like Tylenol® (acetaminophen) or Advil® (ibuprofen). 6
Cost considerations
There can be significant cost variations for muscle relaxant medications, especially as we look at single versus combination products. For example, orphenadrine (e.g., Norflex® and other brands) is a widely used skeletal muscle relaxer. Used in combination with aspirin and caffeine, orphenadrine has been a long-established treatment option as a muscle relaxant.
Orphengesic® Forte is one such combination product. It is indicated for the relief of mild to moderate pain of acute musculoskeletal disorders with rest, physical therapy and other measures. 7 It is marketed under the trade name Norgesic Forte and generic name orphenadrine citrate/aspirin/caffeine by multiple manufacturers. 8
Some of these products can have a very high average ingredient cost. The highest cost product, made by Ina Pharmaceuticals, costs up to $3,055 per prescription. The Ina product is positioned as a generic product and therefore has the majority of utilization (98% of claims).
In contrast, the cost for most generic single agent orphenadrine products is under $60 — less than 2% of the combination products.
This graphic shows a list of orphenadrine, aspirin and caffeine combination products available on market, with their prices:
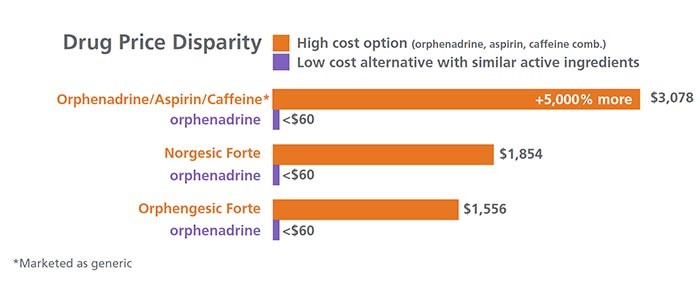
This graphic compares the cost of combination products with orphenadrine, aspirin, and caffeine against the commonly available generic component, orphenadrine. In all three cases orphenadrine is priced at $60. In the first example the combination product (marketed as Orphenadrine/aspirin/caffeine) price is $3,078, which is +5,000% higher. In the second example, the combination product (Norgesic Forte) price is $1,854, and in the third case the combination product (Orphengesic Forte) price is $1,556.
[Chart data compiled by Optum Rx.]
Optum Rx® Vigilant Drug ProgramTM
Optum Rx uses the Optum Rx Vigilant Drug Program to manage combination orphenadrine, aspirin, and caffeine products. The Vigilant Drug Program routinely monitors the potential impact of products like these to protect clients against their high cost.
The High-Cost Generics module of the Vigilant Drug Program is one effective way to deter these kinds of products. By steering members away from high-cost products when lower cost options are available, clients can see significant savings — without compromising the quality of patient care.
For example, effective July 1 of this year, the high cost/high utilization generic product made by Ina Pharmaceuticals has been excluded from coverage since it can easily be replaced at a far lower cost.
Already, brand exclusions in this category have realized savings over $4.6 million during the first three quarters of 2021.
Across our book of business, the Optum Rx Vigilant Drug Program has shown an average of $5.75 PMPM savings when chosen in its entirety. 9, 10
Holistic approach
Optum Rx takes a holistic approach to promoting safety and managing pharmacy costs associated with skeletal muscle relaxants. In addition to the Optum Rx Vigilant Drug Program, Optum Rx also offers the Opioid Risk Management and Utilization Management to meet those goals.
The combination of these programs provides members and clients a comprehensive way to manage the use of skeletal muscle relaxants.
Be sure to ask your consultant or Optum Rx representative about how you can save with the Optum Rx Vigilant Drug Program.
Related content

How a client avoided $109M in pharmacy costs

Drug pricing spotlight: Wound care

How to eliminate prescription waste
Sources
- U.S. Food & Drug Administration. New Drug Therapy Approvals 2020. Current as of January 8, 2021. Accessed October 14, 2021.
- Pain Medicine. Utilization Patterns of Skeletal Muscle Relaxants Among Commercially Insured Adults in the United States from 2006 to 2018. Published March 4, 2021.
- 13.7M estimate extrapolates from previous study based on 67% of US population aged 18-64 in 2019. Annie E. Casey Foundation. Adult population by age group in the United States. Updated September 2020.
- Penn Medicine News. Long-term Use of Muscle Relaxants Has Skyrocketed Since 2005. Published June 25, 2020. Accessed August 18, 2021.
- JAMA Network Open. Assessment of Physician Prescribing of Muscle Relaxants in the United States, 2005-2016. Published June 24, 2020.
- Penn Medicine News. Long-term Use of Muscle Relaxants Has Skyrocketed Since 2005. Published June 25, 2020. Accessed August 18, 2021.
- Galt Pharmaceuticals. Orphengesic Forte Prescribing Information. Accessed August 18, 2021.
- National Drug Code Directory. Accessed July 2021.
- Optum Rx Commercial Direct Client Data, January – December 2020.
- Lists can be implemented as an exclusion or prior authorization strategy. Prior authorization vs. exclusion strategy is not available to clients on the Premium formulary.
STATEMENT REGARDING FINANCIAL INFLUENCE:
This article is directed solely to its intended audience about important developments affecting the pharmacy benefits business. It is not intended to promote the use of any drug mentioned in the article and neither the author nor Optum Rx has accepted any form of compensation for the preparation or distribution of this article.


