Falling behind the guidance
Despite approval of more than 40 new diabetes treatment options since 2005, plus new guidelines and treatment algorithms, the percentage of patients with an A1c over 9% went up between 1999 and 2014.*
The American Diabetes Association blames therapeutic inertia — a hesitancy to change course when treatment falls short of its goals.
Treatment for any chronic disease is admittedly complex. After making a diabetes diagnosis, a physician must weigh 15 potential medications, 30 risk factors, insurance coverages and costs — too many variables to keep in one clinician’s head.
That’s why practices fall back on prescribing old standbys.
Technology can make it easier to keep treatments current by providing timely point-of-care information.
Unfortunately, many decision support solutions are intrusive, interrupting workflows with EHR (electronic health record) alerts. The guidance they offer is general, lacking clinical context. And slow responses leave physicians working with stale information.
Designing decision support
At the Optum Center for Advanced Clinical Solutions, we wanted to know if a user-centered design (UCD) process could help create a better decision support tool.
We built a prototype standards-based medication recommender app for Type 2 diabetes mellitus. Next, we evaluated the app in a simulated setting for effectiveness, efficiency and user satisfaction.
To develop the tool, we interviewed eight clinical leaders with eight rounds of iterative user testing and two to eight prescribers in each round. Then we conducted a validation study with 43 participants. The participants were assigned to one of two groups and asked to complete a two-hour remote user testing session. Both groups reviewed mock patient facts and ordered diabetes medications.
The traditional group first used a mock EHR followed by the prototype app, while the tool group used the prototype app both times.
Users then completed the Kano Survey, the NASA Task Load Index and the System Usability Scale (SUS) questionnaire.
Infographic
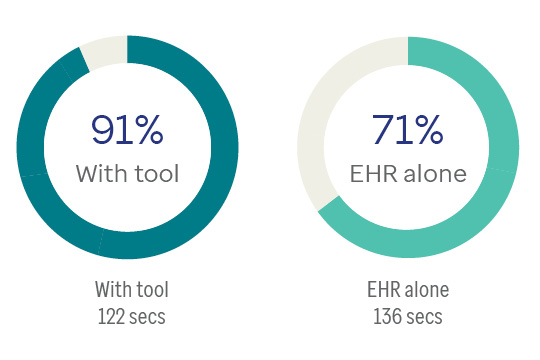
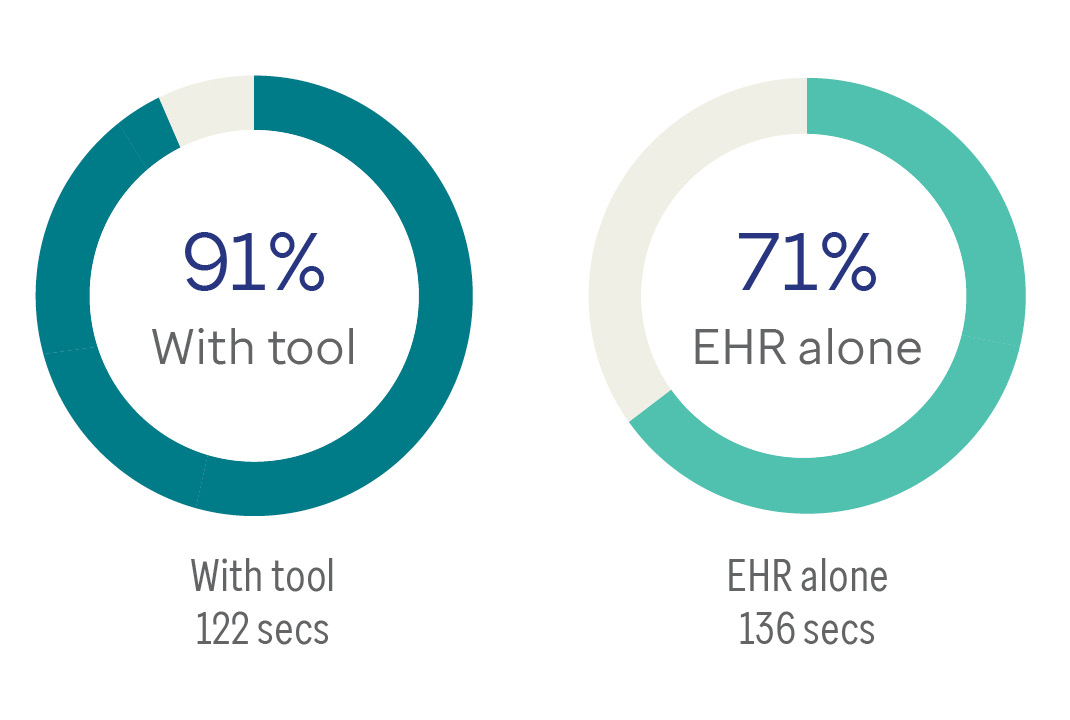
Percentage of orders in adherence with ADA guidelines
Use of the recommender app lowered task time in completing medication orders by 20%.
Percentage of orders in adherence with ADA guidelines
Use of the recommender app lowered task time in completing medication orders by 20%.
Better adherence — and happier clinicians
Across time periods, adherence to evidence-based guidelines was greater with the use of the prototype app than with the EHR. SUS scores also point to high satisfaction.
The results demonstrate that a user-centered design process can play a valuable role in creating point-of-care medication recommenders that hold real promise for improving adherence to ADA guidelines.
As the study suggests, clinicians respond well to decision-support tools that give them what they truly want: a sophisticated, consumer-quality experience that delivers actionable information in real time, presented in a whole-picture context.
By helping physicians overcome treatment inertia, such tools can lead to improved disease management, fewer complications and hospitalizations, and better patient outcomes overall.
Infographic
The prototype T2DM app
Our recommender app gives clinicians patient data on A1c, heart disease, heart failure, kidney disease and other medical problems.


The prototype T2DM app
Our recommender app gives clinicians patient data on A1c, heart disease, heart failure, kidney disease and other medical problems.
