Aduhelm™ for Alzheimer’s: Pipeline implications
Aggressive FDA stance may affect new drugs across classes
Aduhelm™ (aducanumab) is a new drug recently approved by the U.S. Food and Drug Administration (FDA) to treat Alzheimer’s. While Aduhelm is the first therapy that targets the fundamental physiology of the disease, it is not a cure and may only slow disease progression.1
In this article, we examine several ways Aduhelm’s approval could affect the broader drug market for Alzheimer’s and other neurologic conditions, such as Huntington’s disease and Parkinson’s disease.2
While the future of Aduhelm is unclear, we know Alzheimer’s will dominate the pharmacy landscape for years to come so it is important to review potential impacts stemming from the Aduhelm decision.
What’s new is what counts as success.
Aduhelm was approved through an FDA accelerated approval process. This approach allows a drug to be approved if it demonstrates an effect on a surrogate endpoint. A surrogate endpoint is a marker (e.g., laboratory measurement, radiographic image) that is believed to be reasonably likely to predict a clinical benefit. This pathway can be used when measurement of a drug’s effect is lengthy.3
However, not all surrogate endpoints are equally useful.
Savitha Vivian, senior vice president of clinical and formulary services at Optum Rx, notes that validated surrogated endpoints are routinely used in drug approvals without controversy. That’s because they have undergone testing and are accepted by the FDA as evidence of a drug’s benefit.
But the Accelerated Approval program also uses reasonably likely surrogate endpoints. These may stand in for clinical outcomes but are not yet validated. According to Ms. Vivian, “The big issue with Aduhelm is that the surrogate marker used in the trials has not been proven to correlate with a clinically meaningful outcome yet.”
Available clinical trial data have not demonstrated conclusively that Aduhelm slows cognitive decline, which is the hallmark of Alzheimer’s.4 Instead, Aduhelm showed an ability to impact a reasonably likely surrogate endpoint by reducing clumps of protein in the brain called amyloid beta plaques. Some researchers think amyloid plaques cause Alzheimer’s (i.e., the amyloid hypothesis), while others have questions regarding causality.5
Now researchers are wondering whether the endpoints approach used to approve Aduhelm sets a precedent. Can other developers take a similar approach? 6
Beyond Alzheimer’s?
Finding effective treatments to treat neurodegenerative diseases such as Huntington’s disease and Parkinson’s disease has been challenging. In both cases, available treatments can only alleviate symptoms of the disease, but not slow its progression.7, 8
Lack of success has led several pharmaceutical companies to end their neuroscience programs for brain diseases.9
Now the FDA is saying the accelerated approval pathway for Aduhelm is a model they hope can be replicated with neurodegenerative diseases such as Parkinson’s and Huntington’s disease.10
Some researchers would welcome a revised FDA approach, arguing that as long as they can demonstrate some clinical benefit, it’s worth making the treatment available. Others are unsure. One remarked that he was not interested in having multiple drugs, but in having multiple drugs “…that work.”11
This expanded approval pathway from the FDA could open the door to new treatments for substantial patient populations. By itself, Parkinson’s disease is the second most prevalent neurodegenerative disease after Alzheimer’s, affecting approximately one million people in the U.S.12
Rapid new approvals?
If Aduhelm signals a revival of the amyloid hypothesis for Alzheimer’s, it would represent a major shift. In fact, multiple drug makers had decided to drop their Alzheimer’s research programs entirely in recent years.13
According to some researchers, the decision to move on from the amyloid approach came none too soon. By this view, once amyloid became the target, alternative approaches were abandoned, or at least found it difficult to find funding. At least until June of this year, this meant 30 years and billions of dollars wasted.14
But several anti-amyloid drugs remain in trials. Within weeks of the Aduhelm decision, the FDA granted breakthrough therapy designation to two such compounds: Eisai and Biogen’s lecanemab and Eli Lilly’s donanemab.15
Both of these drugs have experienced similarly mixed trial results as Aduhelm. They both saw some reduction of cognitive decline, with varying levels of significance. However, both were quite successful at reducing the amount of amyloid plaque in the brain.16, 17
Roche also has an amyloid reducing candidate called gantenerumab. That drug failed a phase 3 study back in 2014 when it could not demonstrate improved clinical outcomes. But now it’s being tested again specifically for effectiveness at removing amyloid plaques.18
All three of these drugs are currently in phase 3 trials. Trial results are expected by the end of September 2022 (lecanemab) or during 2023 (donanemab, gantenerumab).19, 20, 21
Experts believe that evidence of amyloid-lowering activity might now be enough for these to gain approval – regardless of their effect on cognition.22
The question now is whether potential profits stemming from the Aduhelm approval will draw a host of drug makers into the amyloid space. If the doubts about amyloid reduction prove true, the result might be that promising new development avenues could be squeezed out, while companies do nothing more than multiply the number of drugs that “don’t work.”
How serious is the amyloid comeback?
The three drugs just mentioned were all well advanced in development quite independent of Aduhelm. Is there any reason to believe that the Aduhelm decision will cause a sudden pivot back toward the amyloid hypothesis? This seems unlikely.
It’s worth noting that anti-amyloid trials have been failing to show clinical benefit for years. These failures have triggered a revamped pipeline filled with many alternative approaches to Alzheimer’s treatment.23 A 2020 review of clinical trials over the previous five years showed a growing emphasis on non‐amyloid targets.24
Looking at trials currently underway, there were 28 agents in phase 3 trials as of January 2021. Of these, 17 are disease modifying therapies (DMTs) that are aimed at changing the course of the disease, instead of treating symptoms. Five of these DMTs (29%) have amyloid as their target.25
Stepping further back in the pipeline to phase 2 studies, we see the amyloid activity fall off steeply. There are 74 agents currently in phase 2 trials; 64 of which are potential DMTs. Of the 64 DMTs, only 11 of them aim at amyloid reduction – 17% of the total.26
It’s worth noting that the biopharma industry is investing heavily in many of the new Alzheimer’s approaches. Of the phase 3 trials currently underway, 61% are sponsored by the biopharma industry (as opposed to academic, government, or others).27
This graph shows the wide discrepancy between the trials aimed at amyloid reduction versus other approaches:
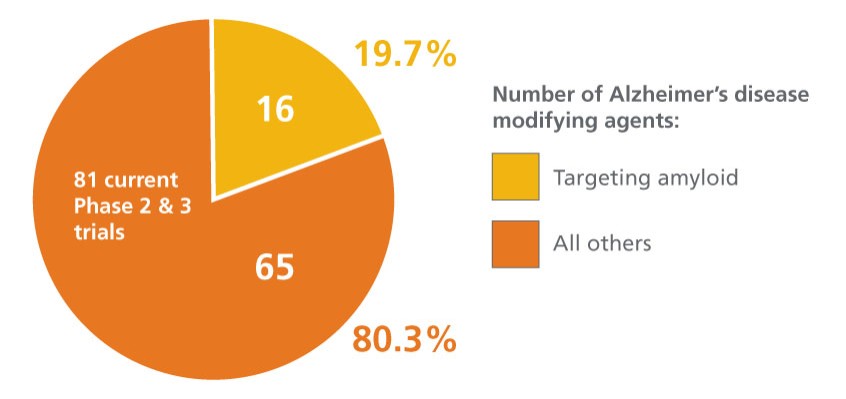

Of the Alzheimer's disease modifying agents currently in clinical trials, 19.7% target the amyloid.
Pie chart shows of the 81 Alzheimer's disease modifying agents currently in phase 2 or 3 clinical trials, 16 or 19.7% target the amyloid.
Source: Alzheimer’s & Dementia: Translational Research & Clinical Interventions. Alzheimer's disease drug development pipeline: 2021. First published May 25, 2021.
Given the controversy and uncertainty around Aduhelm, drug makers will presumably be anxious to see if these newer approaches demonstrate superior results. If so, they would have little incentive to keep investing in anti-amyloids.
Implications
The reason we’re having this discussion is the FDA’s belief that Aduhelm can target and affect the underlying disease process of Alzheimer’s.28 A lot is riding on their judgement.
In the short term, it appears that at least some people will be treated with Aduhelm, at high cost. Drug makers fortunate enough to have their own anti-amyloid candidates already in late stage trials could be poised to profit from this new market, assuming a more forward-leaning FDA position remains in place.
Longer term, we have documented the increasing diversity of targets and therapeutic mechanisms in development right now. We can expect intense interest around the results from the 81 disease modifying therapies candidates currently moving through phase 2 and 3 trials that do not rely on amyloid reduction.
This more diversified drug development pipeline may yield effective new treatments against Alzheimer’s, which would have the potential for significant market penetration. Moving forward, it is reasonable to expect overall spending in the class to rise.
Optum Rx will continue to monitor this important category and keep you informed.
Related content

Is an Effective Alzheimer's Treatment Coming?


Coordination of Benefits (COB) Cost Avoidance
Sources
- FDA News Release. FDA Grants Accelerated Approval for Alzheimer’s Drug. Posted June 7, 2021. Accessed July 9, 2021.
- Nature. Controversial Alzheimer’s drug approval could affect other diseases. Published June 29, 2021. Accessed July 9, 2021.
- FDA. Development & Approval Process: Drugs. Published October 28, 2019. Accessed July 9, 2021.
- BioSpace. Aduhelm Saga Continues as Lawmakers Plan to Investigate Approval, Pricing. Published June 28, 2021. Accessed July 13, 2021.
- Nature. Landmark Alzheimer’s drug approval confounds research community. Published June 8, 2021. Accessed July 9, 2021.
- Nature. Controversial Alzheimer’s drug approval could affect other diseases. Published June 29, 2021. Accessed July 14, 2021.
- ibid.
- Mayo Clinic. Huntington's disease/Treatment. Accessed July 15, 2021.
- Nature. Controversial Alzheimer’s drug approval could affect other diseases. Published June 29, 2021. Accessed July 14, 2021.
- ibid.
- ibid.
- Parkinson Foundation of the National Capital Area. About Parkinson’s Disease. Accessed July 15, 2021.
- MedPage Today. 40 Years of Alzheimer's Research Failure: Now What? Published September 13, 2018. Accessed July 12, 2021.
- STAT News. After amyloid failures, it’s time to take a new tack for treating Alzheimer’s. Published April 30, 2019. Accessed July 15, 2021.
- GlobalData Healthcare. Anti-amyloid therapies signal potential for early Alzheimer’s disease. Published June 30, 2021. Accessed July 12, 2021.
- Alzheimer’s News Today. Lilly’s Donanemab Granted FDA Breakthrough Therapy Status. Published July 1, 2021. Accessed July 12, 2021.
- Alzheimer’s News Today. With New Trial Data, Lecanemab Wins FDA Breakthrough Status. Published June 29, 2021. Accessed July 12, 2021.
- UPDATE: Roche dismisses early filing chatter for its Alzheimer's drug as speculation, remains focused on phase 3 trial. Published July 1, 2021. Accessed July 12, 2021.
- Alzheimer’s News Today. With New Trial Data, Lecanemab Wins FDA Breakthrough Status. Published June 29, 2021. Accessed July 12, 2021.
- Biopharma Dive. Lilly lays out 2-year plan to gain approval for Alzheimer's drug. Published March 15, 2021. Accessed July 15, 2021.
- UPDATE: Roche dismisses early filing chatter for its Alzheimer's drug as speculation, remains focused on phase 3 trial. Published July 1, 2021. Accessed July 12, 2021.
- Nature. Landmark Alzheimer’s drug approval confounds research community. Published June 8, 2021. Accessed July 9, 2021.
- Biomedicines. Reasons for Failed Trials of Disease-Modifying Treatments for Alzheimer Disease and Their Contribution in Recent Research. Published online December 9, 2019. Accessed January 8, 2020.
- Alzheimer's & Dementia: Translational Research & Clinical Interventions. Alzheimer's disease drug development pipeline: 2020. Published July 16, 2020. Accessed January 8, 2020.
- Alzheimer’s & Dementia: Translational Research & Clinical Interventions. Alzheimer's disease drug development pipeline: 2021. First published May 25, 2021. Accessed July 13, 2021.
- ibid.
- ibid.
- FDA News Release. FDA Grants Accelerated Approval for Alzheimer’s Drug. Posted June 7, 2021. Accessed July 9, 2021.
STATEMENT REGARDING FINANCIAL INFLUENCE:
This article is directed solely to its intended audience about important developments affecting the pharmacy benefits business. It is not intended to promote the use of any drug mentioned in the article and neither the author nor Optum Rx has accepted any form of compensation for the preparation or distribution of this article.


