Streamline your clinical research
Clinical research is an essential element of our healthcare system. Without it, we cannot ensure the safety and efficacy of new therapies and major advances in health care technology. But many institutions conducting clinical research face a costly, onerous process.
With Optum clinical trial solutions, researchers can model the effects of inclusion and exclusion criteria to confirm they’re looking at a recruitable population. And they can take things a step further by working with Optum staff to accelerate patient enrollment.

Clinical trial design solutions
Optimize Feasibility and Protocol Development
Reduce the Burden on Clinical Trial Sites
Streamline Data Collection in Clinical Trials
Boost trial impact by finding the right patients
Reach diverse patients with the Optum® Digital Research Network, which offers access to 50 million potential trial participants in the U.S.
Boost trial impact by finding the right patients
Reach diverse patients with the Optum® Digital Research Network, which offers access to 50 million potential trial participants in the U.S.
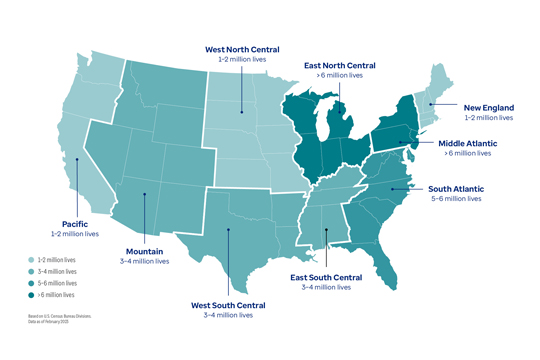
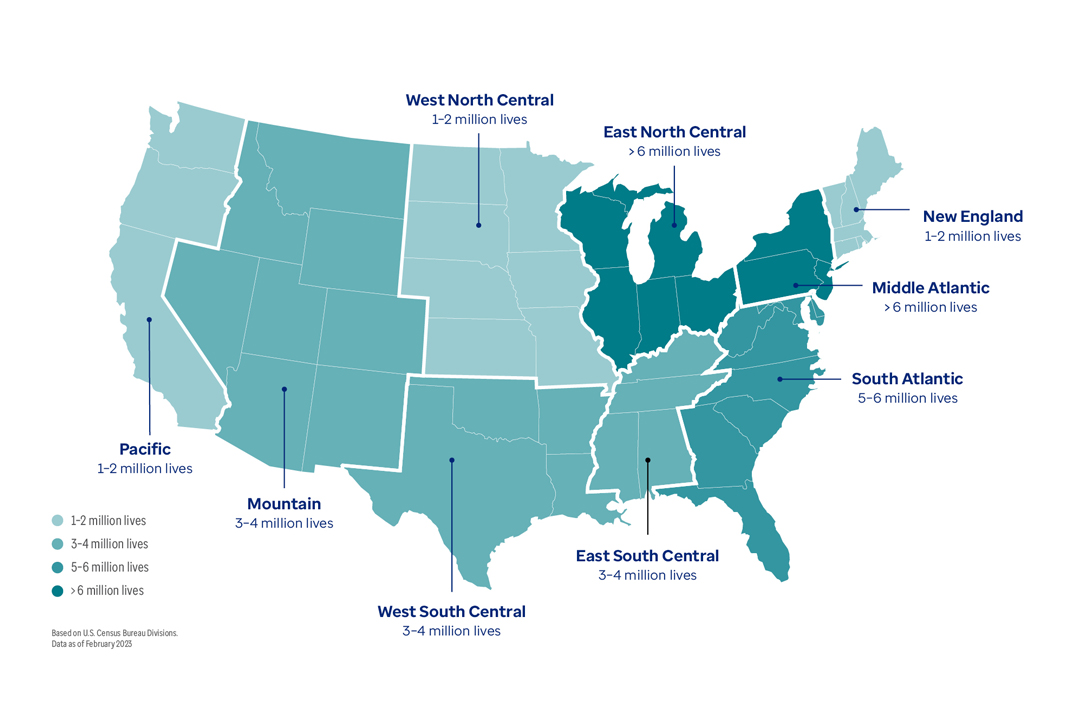
Map of the contiguous United States showing the number of patient lives generated by the Optum Digital Research Network (DRN) in each region, using colored shading to indicate which areas of the country have more and fewer patient lives in the DRN. The breakdown by regions are as follows: the Pacific region has 1–2 million lives, Mountain has 3–4 million lives, West South Central has 3–4 million lives, East South Central has 3–4 million lives, South Atlantic has 5–6 million lives, Middle Atlantic has more than 6 million lives, New England has 1–2 million lives, East North Central has more than 6 million lives, and West North Central has 1–2 million lives. These regions are based on the U.S. Census Bureau divisions. The DRN patient counts in each region are as of February 2023.
Related healthcare insights
Article
Pragmatic trials look increasingly attractive to researchers, but they’re tough to execute. Learn how to simplify the process.
Video
Watch clinical trial leaders from Optum and Biogen discuss how to address trial recruitment struggles by unlocking real-world data (RWD).
Article
Clinical trial protocol design and feasibility often occur in silos. Learn how to make the overall trial design process more effective with RWD and other tools.



